|
6/18/2023 0 Comments Hydrogen atomic mass
For average atomic mass, the isotopes of an element are found in difference abundances or amounts in nature. The difference between the two types of averages is that an arithmetic average are of equal importance, but a weighted average takes into account the importance of some values over others. But for average atomic mass we're calculating a weighted average. It we wanted the arithmetic average then it would be 5.5 because (5+6)/2 = 5.5. Also, scientists needed a pure isotope to base the system on. Scientists used carbon-12 because no other atom has exact whole-number masses in the amu scale. This is why the mass of carbon-12 is 12 amu, not 12.09. Some of the mass of the atom gets converted into binding energy, so the mass of the entire nucleus would actually be less than the mass of each individual proton and neutron added together. 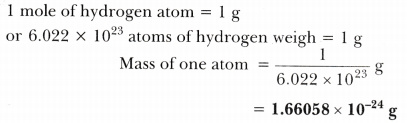
Because of energy-mass equivalence (E = mc^2), we know that energy and mass are interchangeable. Binding energy is the energy that holds the nucleus together. Since the nucleus is full of protons, which has positive charge, they would repel and the nucleus would fly apart. The missing mass is called mass defect, and it represents the binding energy. Following this logic, the carbon-12 atom's atomic mass should actually be 12.09 amu however, the mass is exactly 12 amu. So the mass of a proton is around 1.008 amu, not 1. The relative abundance of Deutrium (1 proton, 1 neutron) is so small that it is barely accounted for when calculating the average atomic mass. Protium is by far the must abundant isotope of hydrogen, and it only contains 1 proton, and no neutrons. The average atomic mass for hydrogen is actually around 1.008 amu. But after isotopes were discovered (oxygen-17, oxygen-18, etc), everything got really confusing, so scientists agreed to use carbon-12. This article was written for you by Samantha, one of the tutors with SchoolTutoring Academy.Good question that requires a kind of long explanation!Ī long time ago, the standard for measuring average atomic mass was actually based on oxygen, and scientists thought all oxygen was oxygen-16 (8 protons, 8 neutrons). Looking to get ready for the ACT? We can help with ACT Prep Therefore, you have 136.2652 g of calcium. Since the atomic mass of calcium is 40.078 amu, the molar mass of calcium is 40.078 g/mole.Ģ) Multiply the number of moles of calcium you have by the molar mass. Therefore, you have 0.4996 mole of nitrogen.Įxample: If you had 3.4 moles of calcium, how many grams of calcium do you have?ġ) Figure out what the molar mass of calcium is. Since the atomic mass of nitrogen is 14.01 amu, the molar mass of nitrogen is 14.01 g/mole.Ģ) Divide the amount of nitrogen you have by the molar mass. 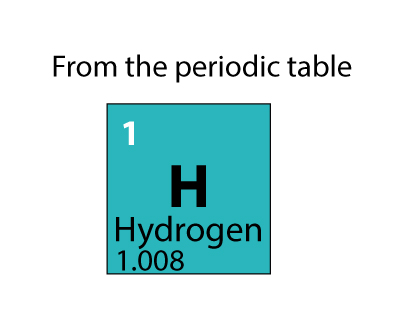
The molar mass of water would then be 18.016 g/mole.Įxample: If you had 7 g of nitrogen, how many moles of nitrogen do you have?ġ) Figure out what the molar mass of nitrogen is. To get the molar mass of water, you would add up the molar masses of 2 hydrogens (2 x 1.01 g) and 1 oxygen (1 x 16 g). For example, a water molecule is made up of 2 hydrogen atoms and 1 oxygen atom. 
To get the molar mass for a compound, you add up all the molar masses of the atoms in the compound. Thus, the molar mass of hydrogen is 1.01 g/mole. This means that 1 mole of hydrogen is 1.01 g. Thus, 1 mole of any element has a mass in grams that is numerically equivalent to its atomic mass.įor example, the atomic mass of hydrogen is 1.01 amu. 12 grams is equal to 1 mole of carbon-12, which has 6.02×10 23 atoms. If we look at 1 atom of carbon-1, it has a mass of 12 amu. Molar mass is used to convert moles to grams.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
